24.1 O2 molecules * 2 = 48.2 Oxygen atoms in 4 moles of O2 As a final bonus calculation, you can convert between moles and mass. On the periodic table, each element has an atomic mass written next to it; Hydrogen's atomic mass is 1.01. This number means two things. First, it tells you the mass of a single atom. For example, 24 grams of 12C atoms would be equal to two moles since 24 grams. To understand how molar mass and Avogadro's number act as conversion.
Chemistry | DIY Projects | Atoms and Atomic Structure
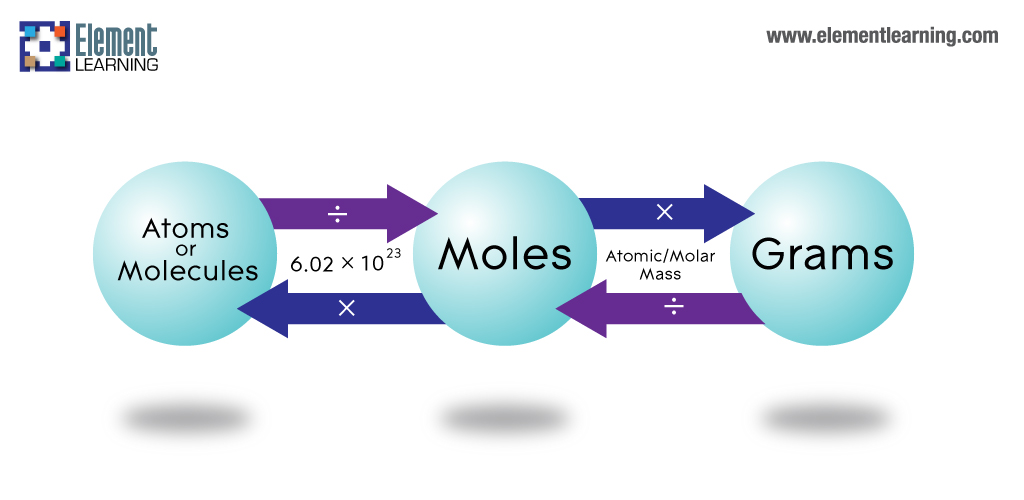
To convert grams into atoms, you have to convert them into molesfirst. Get the molar mass and multiply it by the number of moles toget the atoms.
Grams of mercury to Atoms of mercury?
How do you convert 2.2 grams in to atoms in nitrogen?
How do you convert from grams to atoms and from mass to atoms?
How do you convert grams to atoms with the avogadro's number?
How many Cs atoms are there in a 53.7 gram sample of Cs?
How do you convert from mass to atoms?
How many grams are there in 2.3 x 1024 atoms of silver?
How do you convert moles of atoms to grams?
What is the mass of atoms in grams of 1.02X10 to the 24 atoms Mn?
How do you convert 6.21021 uranium atoms to grams?
What is the mass in grams of 2.01 x 1022 atoms of sulfur?
How do you convert 50.0 grams into atoms?
How many dozens of copper atoms in a penny?
How many atoms are in a sample of 85.979 g of iodine?
Convert 3.55 grams of potassium to atoms?
How many atoms are in 1.500 kilograms of gold?
How do you convert atoms to grams with Avogadro's number?
What would you multiply atoms of germanium by to get the units grams of germanium?
What would you multiply atoms of boron by to get the units grams of boron?
What would you multiply grams of oxygen by to get the units atoms of oxygen?
What relationships are needed to convert 1.5 grams of carbon to the number of carbon atoms?
What has more atoms 2.88 grams of helium atoms or 16.4 grams of zinc atoms?
What is the mass of 1.505 x 10²³ carbon atoms?
How many atoms are in one gram of 13Al?
How do you find the mass of a sample of 1.72 x 1023 atoms of potassium?
How do you convert grams to cups?
Convert grams to ounces?
How do you convert 2.11x10 to the 24 atoms of copper to mass in grams?
How many moles of atoms are contained in 22.6 g of the Co?
What is the total number of atoms contained in 80 grams of neon?
How many atoms are present in 400g Iron?
How do you convert 300 grams into cups?
How do you go from atoms to grams?
How many atoms of Mercury in 65 grams of Mercury?
How many atoms of gold are in 5 grams of gold?
How do you convert grams into millograms?
How many atoms are in 155 grams of calcium?
How do you convert atoms to molecules and molecules to atoms?
Which has the greatest number of atoms- 3.5 grams of helium or 3.5 grams of lithium?
How do you convert the mass of one hydrogen atoms in grams?
Convert 100 grams of butter to cups?
How do you convert 200 kg into grams?
How do you convert 21 grams to Mlli grams?
How do you convert 6 kilograms to grams?
What is the mass in grams of 2x1012 atoms of potassium?
How many atoms are in 25.1 grams in sulfur?
How can you convert 79 grams to pounds?
How many atoms are in 1.9 g Carbon?
How do you convert mass into moles of an element?
How do you convert grams to atoms?
Avogadro's number is one of the most important constants used in chemistry. It is the number of particles in a single mole of a material, based on the number of atoms in exactly 12 grams of the isotope carbon-12. Although this number is a constant, it's experimentally determined, so we use an approximate value of 6.022 x 1023. So, you know how many atoms are in a mole. Here's how to use the information to determine the mass of a single atom.
Avogadro's Number Example Problem: Mass of a Single Atom
Question: Calculate the mass in grams of a single carbon (C) atom.
Solution
To calculate the mass of a single atom, first look up the atomic mass of carbon from the periodic table.
This number, 12.01, is the mass in grams of one mole of carbon. One mole of carbon is 6.022 x 1023 atoms of carbon (Avogadro's number). This relation is then used to 'convert' a carbon atom to grams by the ratio:
This number, 12.01, is the mass in grams of one mole of carbon. One mole of carbon is 6.022 x 1023 atoms of carbon (Avogadro's number). This relation is then used to 'convert' a carbon atom to grams by the ratio:
mass of 1 atom / 1 atom = mass of a mole of atoms / 6.022 x 1023 atoms
Plug in the atomic mass of carbon to solve for the mass of 1 atom:
mass of 1 atom = mass of a mole of atoms / 6.022 x 1023
mass of 1 C atom = 12.01 g / 6.022 x 1023 C atoms
mass of 1 C atom = 1.994 x 10-23 g
mass of 1 C atom = 1.994 x 10-23 g
Answer
The mass of a single carbon atom is 1.994 x 10-23 g.
Applying the Formula to Solve for Other Atoms and Molecules
Although the problem was worked using carbon (the element upon which Avogadro's number is based), you can use the same method to solve for the mass of an atom or molecule. If you're finding the mass of an atom of a different element, just use that element's atomic mass.
If you want to use the relation to solve for the mass of a single molecule, there's an extra step. You need to add up the masses of all of the atoms in that one molecule and use them instead.
Let's say, for example, you want to know the mass of a single atom of water. From the formula (H2O), you know there are two hydrogen atoms and one oxygen atom. You use the periodic table to look up the mass of each atom (H is 1.01 and O is 16.00). Forming a water molecule gives you a mass of:
1.01 + 1.01 + 16.00 = 18.02 grams per mole of water
and you solve with:
mass of 1 molecule = mass of one mole of molecules / 6.022 x 1023
mass of 1 water molecule = 18.02 grams per mole / 6.022 x 1023 molecules per mole
mass of 1 water molecule = 2.992 x 10-23 grams
